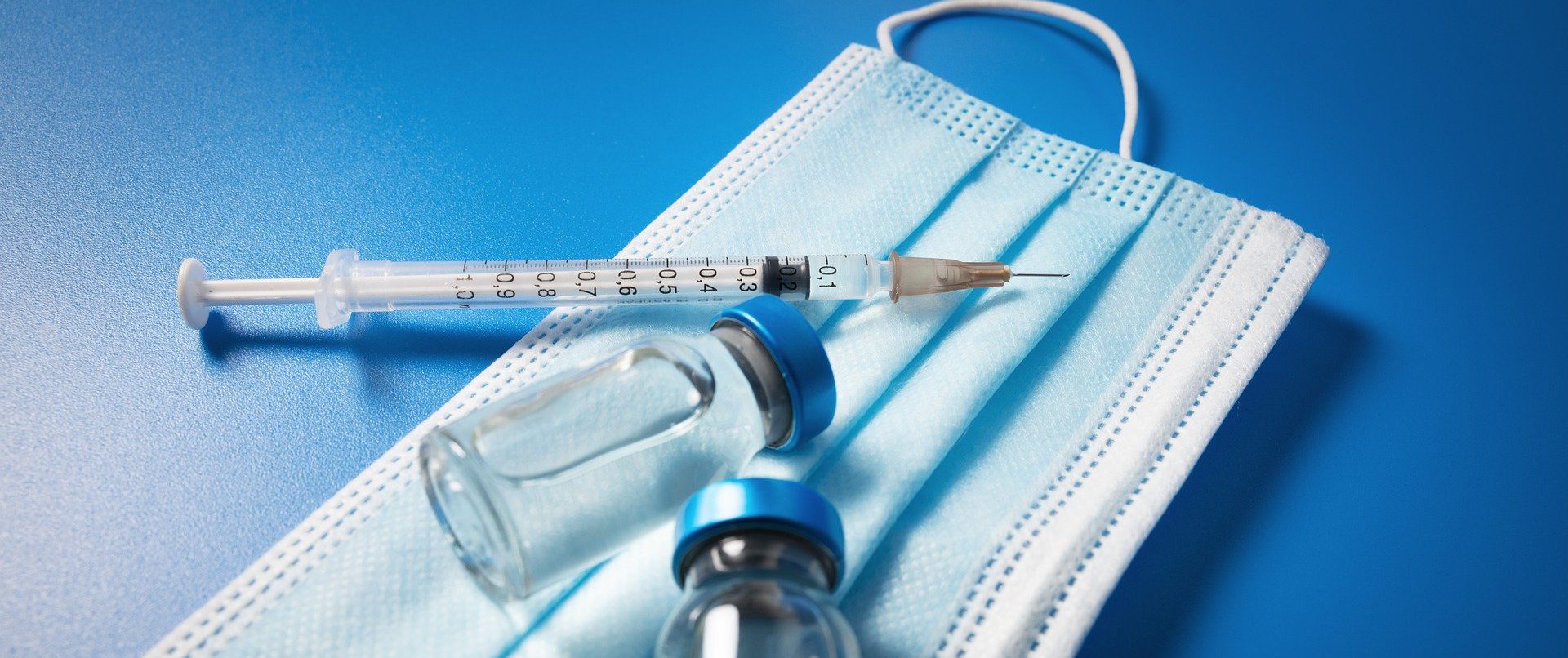
Best practices as recommended by WHO and United States Pharmacopoeia are being implemented for performing tests on therapeutic goods.
Recent Updates
- Requirements for Grant Of Drug Manufacturing License by the Way of Basic & Semi-Basic Manufacturing for Blood/ Plasma EstablishmentsMarch 30, 2026
- Notification (S.R.O. 515(l)/2026)March 30, 2026
- Invitation to bid (For the supply of chemicals, glassware, reference standards/USP standards/CRMs and columns)March 25, 2026
- Decisions of the 123rd meeting of the Medical Devices Board (MDB) held on 25-02-2026 (Manual Applications Part-II)March 25, 2026
- Tender for Provision of Record Management ServicesMarch 25, 2026
- Draft Therapeutic Goods (Import and Export) Rules 2026 (For Comments)March 18, 2026
- 17. Volunatry Recall. Osilex-D (Glitz Pharma)March 11, 2026
- Stakeholders are invited to comment on Draft Post-registration variation guidelines for pharmaceutical and biological products (3rd Edition)March 11, 2026
- 16. Recall Alert (Human) – CDL Karachi declared substandard products.March 11, 2026
- 15. Recall Alert (Veterinary) – DTLs Punjab declared substandard products.March 11, 2026
- Show all updates
Chief Executive Officer
 Dr. Obaidullah
Dr. Obaidullah

E-Services
With the effective use of Information Technology, we have implemented a number of online services and continually working for more. Click here to browse available services.

Guidelines
Regulatory compliance is essential and indispensable in order to assure the safety, quality and efficacy of therapeutic goods. Guidance documents published by the Authority are meant to explain the current interpretation or policy for better understanding of the industry and

Quality Control Labs
For ensuring quality therapeutic goods, a network of quality control laboratories has been established. DRAP approved products and suspect therapeutic goods are tested for their quality in accordance with best international practices. To keep the citizens safe from substandard and

Therapeutic Products Recalls
The Recalled Product Index provides access to consumers, healthcare professionals, industry, hospitals and sale outlets related to the information of recalled action taken for defective therapeutic goods. The following section provides an index of recalled actions in which a public

Health Professional Alerts
DRAP’s issues safety updates and alerts related to the therapeutic goods in order to ensure that healthcare professionals and other stakeholders have timely access to the relevant information on safety, quality and efficacy issues or benefit/risk evaluations of the drugs,

Medicine Availability
The Drug Regulatory Authority of Pakistan (DRAP) is committed to safeguard public health by ensuring the consistent availability of safe, efficacious, and quality medicines nationwide. Recognizing the critical impact of drug shortages, particularly of essential and lifesaving medicines, on patient
Online System for Controlled Drugs Quota Allocation
The Drug Regulatory Authority of Pakistan (DRAP) is pleased to announce the launch of an Online Controlled Drugs Quota Allocation System. The system enables stakeholders to submit electronic applications for quota allocation of controlled drugs in a transparent, efficient, and paperless manner.
Lot Release Application System
We've launched Lot Release Application System. Please use following link to submit your lot release applications for biological products.
Do you know?
DRAP ensures that every drug, medical device or cosmetic, alternative medicine and health product must have a certain standard of quality and is safe and effective for your use. See in links below how it is done and what to do in case you find a suspect therapeutic good.